Clozapine in Psychiatry
1. History / background
- Discovery & use
Clozapine was developed in the 1950s/60s as one of the “atypical” or “second-generation” antipsychotics. Its unique profile emerged when it showed efficacy in patients who had failed conventional antipsychotics, along with a relatively low risk of extrapyramidal side effects (EPS).
However, after reports of agranulocytosis in the 1970s, it was withdrawn or restricted in many countries, then reintroduced later under strict monitoring.
Over time, clozapine became recognized as the “gold standard” for treatment-resistant schizophrenia (TRS) because of superior efficacy in that group (especially for reducing suicidality). - Role in guidelines
Maudsley consistently emphasizes that clozapine should be offered as soon as treatment resistance is apparent, rather than delaying too long — i.e. earlier use of clozapine is associated with better response.
In their algorithm for schizophrenia, after failure of two adequate antipsychotic trials (one should be a second-generation), the next step is to switch to clozapine.
Delaying initiation of clozapine (i.e. waiting too many “failed switches”) is associated with a reduced probability of response. - Evolution of monitoring & safety
Because of risks (especially agranulocytosis/neutropenia, seizure risk, myocarditis/cardiomyopathy, metabolic issues, hypersalivation, etc.), clozapine is used under “specialist monitoring” frameworks (e.g. REMS in the US) and regular blood counts. (U.S. Food and Drug Administration)
2.Mechanism of Action (MOA)
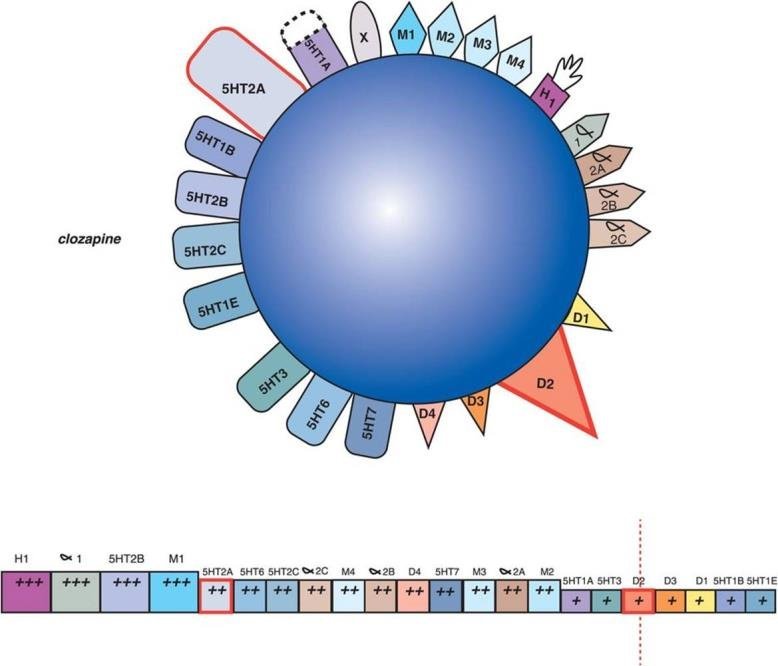
Clozapine’s pharmacology is complex and “dirty” (i.e. hits many receptors). There is no single receptor to which its full clinical effects can be ascribed; rather it is a multi-receptor “balancer.” Key features:
- Dopamine receptors:
It is a relatively weak antagonist at D₂ receptors (lower affinity than many typical antipsychotics), and has a relatively fast off-rate. This may help mitigate EPS.
It also interacts (antagonism) with D₁, D₄ and possibly other dopamine subtypes. - Serotonin receptors:
It antagonizes multiple 5-HT (serotonin) receptors (5-HT₂A, 5-HT₂C, 5-HT₆, 5-HT₇). The antagonism at 5-HT₂A is thought to mitigate some of the dopaminergic blockade side effects, and the 5-HT₂C antagonism may contribute to appetite/weight gain. - Adrenergic receptors:
α₁ and α₂ antagonism contributes to orthostatic hypotension, sedation, tachycardia, etc. - Histaminergic receptor:
H₁ antagonism contributes to sedation and weight gain. - Muscarinic (cholinergic) receptor:
It has antimuscarinic (M₁, M₂, M₃, M₄) effects and some anticholinergic action, which contribute to side effects (dry mouth, blurred vision, constipation) but also may modulate other brain circuits. - Other / glutamatergic modulation:
There is some evidence that clozapine modulates NMDA/glutamate systems, GABA, and possibly nicotinic receptor pathways, which may contribute to its unique efficacy, especially in “treatment-resistant” psychosis and negative/cognitive symptoms.
Because of this pleiomorphic action, clozapine often works when other antipsychotics fail (i.e. it has broader “reach” into brain circuits).
3.Evidence / clinical trials / what supports its use
- In controlled trials and observational studies, clozapine shows superiority over other antipsychotics in treatment-resistant schizophrenia (i.e. patients who do not respond to at least two trials). Maudsley notes that while there are relatively few large, modern randomized controlled trials (RCTs) comparing high vs standard doses, the weight of evidence (including open and controlled studies) support its use in TRS.
- Clozapine has evidence for anti-suicidal efficacy beyond its antipsychotic effect, which is unique among antipsychotics.
- It is often used in augmentation trials (though evidence is weaker) for clozapine non-responders (i.e. those who fail even clozapine monotherapy). Maudsley will caution augmentation strategies as time-limited and carefully monitored.
- Some trials have looked at higher doses, or faster titration, but the designs are generally small and of older vintage. Maudsley tends to emphasize safety over aggressive titration.
Thus, the main “trial support” is in TRS, plus real-world observational data, clinical experience, and meta-analyses.
4.Indications
Approved / core indications
- Treatment-resistant schizophrenia (i.e. failure of at least two antipsychotics, one of which should be a second-generation). Maudsley strongly places clozapine here.
- Psychosis in Parkinson’s disease (some jurisdictions allow very low-dose clozapine for PD psychosis, since it causes minimal motor worsening).
- In some jurisdictions, reduction of suicide risk in schizophrenia (i.e. in suicidal patients).
- Sometimes, “off-label” or more experimental in severe aggression/violent behavior in psychotic patients, particularly if resistant to others (but only in specialized settings).
Maudsley is cautious about off-label use and tends to emphasize that any use of clozapine must weigh benefit vs risk and requires specialist oversight.
5.Initiation / how to start (titration, cross-tapering, monitoring)
This is a critical and high-risk stage. The Maudsley guidelines (and standard practice) emphasize “start low, go slow,” close monitoring, and cross-tapering of prior antipsychotics.
Baseline assessments before initiation
Before starting clozapine, one should obtain:
- Full blood count (FBC) / absolute neutrophil count (ANC)
- Baseline metabolic panel: glucose, lipids, HbA1c
- ECG (for QT prolongation, arrhythmia risk)
- Troponin / CK / CRP (some centers do baseline myocarditis screening)
- Weight, BMI, waist circumference, blood pressure
- Liver and renal function tests
- Uric acid, prolactin, etc (depending on local practice)
- Pregnancy test in women of childbearing potential
- Baseline bowel habit / GI motility history
- Screening for seizures risk, baseline EEG if risk factors
- Baseline ECG, orthostatic BP, heart rate
- Smoking status, concomitant medications (especially those affecting clozapine metabolism)
- Informed consent and explanation of risks (agranulocytosis, myocarditis, seizures, etc.)
Initiation
The usual approach (and what many guideline sources, including ones referenced by Maudsley, recommend) is:
- Day 1: Start 5 mg once or twice (i.e. 12.5 mg QD or BID).
- Then increase cautiously: increments of 25–50 mg/day if tolerated, usually every day or every other day, until a gradual build-up. (PMC)
- By end of week 1 typical total daily dose may reach perhaps 50-100 mg (depending on tolerance).
- Over the next week or two, climb more slowly toward a target dose (often 250–450 mg/day as a starting “therapeutic” range, though many patients may need higher).
- Subsequent increments should be cautious — typically once or twice per week, not exceeding (say) a 100 mg change, and observing for side effects (orthostasis, sedation, seizures, myocarditis).
- Maximal doses are often around 900 mg/day in many jurisdictions; exceeding that is rarely justified given risk. (Psychopharmacology Institute)
- If outpatient/community initiation (i.e. not full in-patient monitoring), a still slower titration is prudent.
Cross-tapering of previous antipsychotics
- Common practice is to maintain the existing antipsychotic at its full dose during the early days of clozapine initiation, while the clinician monitors for side effects and response.
- After clozapine reaches a moderate dose (e.g. ~100–200 mg/day), you may begin to taper the prior antipsychotic gradually — perhaps by ~25 % per week (depending on tolerance, overlap, symptom burden).
- In some cases, more cautious tapering is needed if side effects or interactions are present.
Monitoring during initiation
- Frequent vital signs (BP, HR, orthostatic readings)
- Daily FBC / ANC monitoring (depending on local rules) or per protocol
- Monitor for signs of myocarditis/cardiomyopathy: chest pain, dyspnea, tachycardia, raised CRP/CK/troponin
- Monitor for sedation, seizures, hypersalivation, GI side effects, orthostatic hypotension
- Monitor metabolic parameters periodically
- Monitor plasma clozapine levels (therapeutic drug monitoring) in steady state to guide dosing
6.Managing / tackling side effects (general principles)
Clozapine has a rich side effect profile. Maudsley’s guidance emphasizes vigilance, prevention, and symptom-targeted strategies. Here is a (not exhaustive) list of common and serious side effects and how to manage them:
| Side effect | Mechanism / risk factors | Management / mitigation |
|---|---|---|
| Sedation / somnolence | Histaminergic, anticholinergic | Start at night, divide dosing, reduce dose if possible, adjunct stimulants (with caution) |
| Orthostatic hypotension, tachycardia | α₁ blockade, autonomic reflex | Slow titration, increase salt & fluids, compression stockings, small meals, dose adjustments, sometimes midodrine |
| Excessive salivation (hypersalivation) | Partial stimulation of M3, cholinergic imbalance | Sugar-free gum, atropinic agents (glycopyrrolate, hyoscine), sublingual atropine, metoclopramide patch, consider dose split |
| Constipation / GI hypomotility | Anticholinergic, reduced GI motility (very serious) | See “CIGH” section below (laxatives prophylaxis, high fiber, hydration) |
| Weight gain / metabolic syndrome | 5-HT₂C, H₁ antagonism, metabolic changes | Diet, exercise, monitoring glucose/lipids/HbA1c, early intervention, possibly metformin, switching off agents that worsen metabolic profile |
| Hyperlipidemia / dyslipidemia | Metabolic derangement | Periodic lipid monitoring, statins if needed |
| Hyperglycemia / diabetes | Insulin resistance, weight gain | Regular glucose / HbA1c, lifestyle, treat when needed (metformin, etc.) |
| Seizures | Dose-related risk, lowering seizure threshold | Avoid rapid dose escalation, monitor plasma levels, if risk high use prophylactic antiepileptic (valproate, lamotrigine, low-dose antiepileptic) |
| Myocarditis / cardiomyopathy | Possibly immune / inflammatory mechanism (esp. early in treatment) | Monitor CRP/CK/troponin regularly (weekly for first weeks), ECG, echo if signs, stop clozapine if suspected |
| Neutropenia / agranulocytosis | Bone marrow toxicity | Mandatory FBC / ANC monitoring. If neutrophils drop, follow stopping rules, re-challenge only per protocol |
Maudsley tends to advise using the lowest effective dose, monitoring early and frequently, and carefully weighing augmentation or side-effect-targeted medications only when essential.
Clozapine plasma levels: Maudsley suggests a therapeutic window of ~0.35 mg/L to 0.60 mg/L (i.e. 350–600 ng/mL) as a guide, while recognizing that some patients may require levels outside these bounds and that toxicity signs are more important to heed. (SPS – Specialist Pharmacy Service). If the patient begins or quits smoking, or has severe infection, dose adjustments (often downward) may be required because metabolism (CYP1A2) is affected by smoking/inflammation. (SPS – Specialist Pharmacy Service)
When side effects are emerging, the clinician should (a) consider dose reduction, (b) treat symptoms (e.g. anticholinergics, laxatives) (c) consider switching dosing schedule (e.g. split doses) or (d) stop clozapine if life-threatening side effects occur (e.g. agranulocytosis, myocarditis).
7.Clozapine-Induced Gastrointestinal Hypomotility (CIGH) & Management
One of the perhaps underappreciated but serious risks of clozapine is gastrointestinal hypomotility, which can lead to constipation, ileus, bowel obstruction, even perforation and death. Maudsley is likely to treat this risk with serious caution, recommending prophylaxis and active management.
Pathophysiology
- Clozapine’s anticholinergic / antimuscarinic action slows gut smooth muscle contractility.
- It can reduce GI motility at multiple levels (colon, small intestine), slow transit, reduce peristalsis.
- Patients may not report constipation early (blunted symptoms), so the risk is insidious.
- In severe cases, fecal impaction, bowel obstruction, paralytic ileus, megacolon or even bowel necrosis can occur.
Prevention / prophylaxis
- At the time of starting clozapine, prophylactic laxative therapy is strongly recommended (e.g. stimulant laxatives, osmotic agents).
- Encourage high fibre diet, adequate fluid intake, regular exercise (if medically feasible).
- Monitor bowel habits daily or regularly (e.g. ask patients to report any change, measure bowel frequency).
- Avoid co-prescribing other strong constipating agents (e.g. anticholinergics, opioids) if possible.
- Use “bowel charts” in inpatient settings to ensure monitoring.
- Educate patient and caregivers about red flags (no stool or flatus, colicky pain, abdominal distension, nausea/vomiting).
Monitoring & early detection
- Ask regularly about bowel frequency, stool consistency, pain/discomfort, bloating.
- In suspicious cases, evaluate abdominal exam, abdominal X-ray or CT if obstruction suspected.
- Monitor for signs of ileus (absent bowel sounds, distension, vomiting).
Management when constipation or hypomotility is detected
- Increase or optimize laxative regimen: e.g. stimulants (senna, bisacodyl), osmotic agents (lactulose, polyethylene glycol), prokinetic agents.
- Consider lubiprostone, prucalopride, macrogols, magnesium, others as per local protocol (though evidence is limited).
- In severe cases, bowel cleansing, enemas, disimpaction may be necessary.
- If signs of bowel obstruction or ileus: stop clozapine (or hold), urgent GI workup, possibly surgical consultation.
- Reintroduction of clozapine after severe GI events must be cautious, with continuing prophylaxis.
- Use the lowest effective clozapine dose to reduce GI burden.
- In some cases, adjunct agents targeting motility (e.g. cisapride, prucalopride) may be considered (if locally available and safe).
- Monitor closely for relapse of GI side effects.
Given the high morbidity/mortality associated with severe CIGH, many psychiatrists treat prophylactic laxatives as mandatory with clozapine.
8.Special issues: Rechallenge, cessation, adjusting for interacting factors
- Rechallenge after neutropenia/agranulocytosis: In some jurisdictions, if neutropenia was mild and transient, a monitored rechallenge is possible using stricter monitoring and perhaps using granulocyte-stimulating agents (e.g. G-CSF). Maudsley outlines “restart rules” protocols in their tables.
- Stopping clozapine: Discontinuation must be cautious; abrupt cessation risks rebound psychosis, cholinergic rebound, withdrawal symptoms. Maudsley suggests tapering where possible.
- Adjusting for smoking / infection: Because smoking induces CYP1A2, starting or stopping smoking can change clozapine metabolism significantly; thus, one should monitor plasma levels and adjust dose, typically downward when stopping smoking. (SPS – Specialist Pharmacy Service)
- Drug interactions: Many drugs inhibit CYP1A2, CYP3A4, or CYP2C19, and may increase clozapine levels (e.g. fluvoxamine, ciprofloxacin, oral contraceptives). Maudsley warns to monitor levels and side effects.
- Plasma monitoring: As noted, aim for 0.35–0.6 mg/L (350–600 ng/mL) as a therapeutic window, but interpret levels in context of side effects. (SPS – Specialist Pharmacy Service)
- Long-term monitoring: Even after stabilization, periodic FBC / ANC, metabolic checks, cardiovascular screening, ECG, etc., are required.
9.Practical tips, “pearls & caveats” (from MPG 15th Edition)
- Use the lowest effective dose don’t escalate just for the sake of achieving a “high dose.”
- Titrate slowly to reduce risk of orthostasis, seizures, myocarditis.
- Monitor carefully in the first 4–8 weeks, since many serious adverse effects (neutropenia, myocarditis) occur early on.
- Be extra cautious in older patients, those with cardiac or metabolic comorbidity.
- Use prophylactic laxatives from the start; don’t wait until constipation is severe.
- Always consider drug – drug interactions and changing metabolism (e.g. smoking, infections) when adjusting dose.
- Plasma level monitoring is a useful guide but clinical signs and side effects take precedence (i.e. treat the patient, not the number).
- Have clear protocols for stopping/holding clozapine in the face of serious adverse events (agranul, myocarditis, severe GI complications).
- When a patient is non-responding even on clozapine, augmentation (e.g. ECT, addition of other agents) should be time-limited and carefully supervised.
- Delay in initiating clozapine is associated with poorer odds of response, thus one should not wait excessively after two failed antipsychotics.
10.References
- Taylor, D. M., Barnes, T. R. E., & Young, A. H. (2025). The Maudsley prescribing guidelines in psychiatry (15th ed.). Wiley.
- Howes, O. D., & colleagues. (2023). Improving clozapine utilization will require continued advocacy, drug reform, and clinician reform. Therapeutic Advances in Psychopharmacology. Link ->
Give Your feedback: