GLP-1 Agonists in Psychiatry
Introduction
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are a class of medications initially developed for the treatment of type 2 diabetes mellitus (T2DM) that have garnered significant attention for their efficacy in weight management. Their potential applications in psychiatry stem from their pleiotropic effects, which extend beyond glycemic control and satiety to include direct and indirect actions within the central nervous system (CNS), influencing mood, reward processing, and neuroinflammation.
Historical Context
The story of GLP-1 RAs begins with the discovery of incretins, gut-derived hormones that potentiate glucose-dependent insulin secretion. GLP-1 is one such incretin (from INtestinal seCRETion of INsulin). The term glucagon-like peptide derives from its sequence homology with the hormone glucagon (Gk. glukus “sweet” + agein “to lead”). The first GLP-1 RA, exenatide, was approved for T2DM in 2005, sourced from the saliva of the Gila monster (Heloderma suspectum). The subsequent observation of significant weight loss and emerging data on CNS GLP-1 receptor distribution paved the way for psychiatric investigation.
Mechanism of Action (MOA)
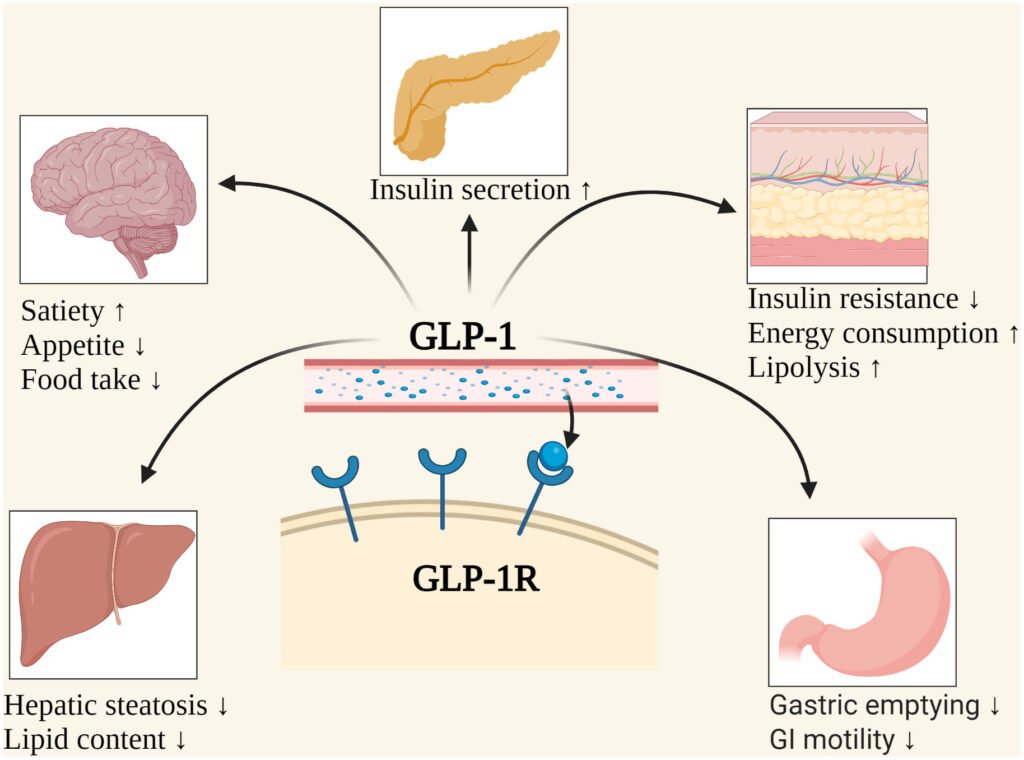
The mechanism of GLP-1 RAs is multifaceted, involving peripheral and central pathways:
- Peripheral Mechanisms:
- Glucose-Dependent Insulin Secretion: They enhance insulin release from pancreatic β-cells in response to elevated blood glucose, minimizing the risk of hypoglycemia.
- Suppression of Glucagon Secretion: They inhibit glucagon release from pancreatic α-cells, reducing hepatic glucose production.
- Gastric Emptying Delay: They slow gastric emptying, contributing to prolonged satiety and reduced postprandial glucose spikes.
- Promotion of Satiety: They act on receptors in the vagus nerve and brainstem to signal fullness.
- Central Nervous System (CNS) Mechanisms (Most Relevant to Psychiatry):
- Neurocircuitry of Reward and Motivation: GLP-1 receptors are densely expressed in key brain regions, including the hypothalamus, ventral tegmental area (VTA), and nucleus accumbens (NAc). Agonism here:
- Modulates Dopaminergic Signaling: Attenuates the dopamine release in the NAc typically associated with the hedonic response to palatable food, drugs of abuse, and potentially other rewarding stimuli.
- Reduces Motivation for Reward: Acts on the mesolimbic pathway to decrease the drive to seek out rewarding substances.
- Neuroinflammation: GLP-1 RAs exhibit anti-inflammatory properties, reducing the release of pro-inflammatory cytokines (e.g., TNF-α, IL-1β) and modulating microglial activation. Chronic neuroinflammation is a postulated pathophysiological mechanism in major depressive disorder and other neuropsychiatric conditions.
- Neuroprotection and Neurogenesis: Preclinical models suggest GLP-1 RAs may promote synaptic plasticity, increase neurogenesis in the hippocampus, and provide protective effects against oxidative stress, akin to mechanisms proposed for some antidepressants.
- Neurocircuitry of Reward and Motivation: GLP-1 receptors are densely expressed in key brain regions, including the hypothalamus, ventral tegmental area (VTA), and nucleus accumbens (NAc). Agonism here:
Clinical Relevance
- Non-Psychiatric Applications:
- T2DM: First-line agents for patients with T2DM and established cardiovascular disease or obesity.
- Obesity: Semaglutide (Wegovy®) and liraglutide (Saxenda®) are FDA-approved for chronic weight management.
- Cardiovascular Risk Reduction: Several GLP-1 RAs have demonstrated cardioprotective benefits, reducing major adverse cardiovascular events.
- Psychiatric Applications (Mostly Off-Label & Investigational):
- Binge-Eating Disorder (BED) and Food Addiction: The most direct psychiatric application. By powerfully reducing obsessive food thoughts and the compulsive drive to binge, GLP-1 RAs can target the core pathophysiology of BED. Example: A patient with severe BED and obesity, who has failed multiple SSRIs and psychotherapy, experiences a dramatic reduction in binge episodes and pre-occupation with food after starting liraglutide, facilitating engagement in behavioral therapy.
- Substance Use Disorders (SUDs): Preclinical data is robust, showing reduced self-administration of alcohol, cocaine, opioids, and nicotine in animal models. Human trials are ongoing. The proposed mechanism is the dampening of the dopamine-driven reward signal in the NAc.
- Major Depressive Disorder (MDD) and Inflammation: The anti-inflammatory and neuroprotective effects position GLP-1 RAs as a novel therapeutic avenue, particularly for the subset of patients with MDD with high inflammatory biomarkers or metabolic comorbidity (“metabolic depression”).
- Neurocognitive Disorders: The neuroprotective properties are being investigated for potential benefits in Alzheimer’s and Parkinson’s diseases, with trials ongoing.
Side Effect Profile
Understanding the side effect profile is crucial for psychiatric patients, who may be more vulnerable to their subjective experience.
- Most Common (Typically Transient):
- Gastrointestinal: Nausea (~20-50%), vomiting, diarrhea, constipation. These are dose-dependent and often wane over weeks. Management: Start low, go slow with titration.
- Decreased Appetite: A therapeutic effect that can be perceived as a side effect.
- Less Common but Serious:
- Pancreatitis: Warning signs include severe abdominal pain.
- Gallbladder Disease: Increased risk of cholelithiasis.
- Hypoglycemia: Rare when used as monotherapy, but risk increases with concomitant insulin or sulfonylureas.
- Diabetic Retinopathy: A signal in one trial with semaglutide; clinical significance is debated.
- Suicidal Ideation: Not identified as a signal in large cardiovascular outcome trials or obesity studies. However, the European Medicines Agency (EMA) is conducting a review of this potential risk, and vigilance is warranted, particularly in patients with pre-existing psychiatric illness.
New Research Findings & Future Directions
- Dual and Triple Agonists: Next-generation agents like tirzepatide (a GIP/GLP-1 dual agonist) and retatrutide (a GIP/GLP-1/glucagon triple agonist) show even more profound efficacy for weight loss and glycemic control. Their psychiatric applications are entirely unexplored but represent a significant frontier.
- Neuropsychiatric Clinical Trials: Numerous Phase II/III trials are actively recruiting to investigate GLP-1 RAs (primarily semaglutide) for conditions including:
- Alcohol Use Disorder
- Binge-Eating Disorder (as a primary endpoint)
- Cognitive Function in Schizophrenia
- Bipolar Depression
- Mechanistic Imaging Studies: fMRI studies are underway to visualize how GLP-1 RAs alter brain activity in reward circuits (e.g., NAc, orbitofrontal cortex) in humans with addictions.
Use in Psychiatry: Practical Considerations
- Current Status: As of now, the use of GLP-1 RAs in psychiatry is off-label and investigational, except for their use in treating obesity, which is often a comorbid condition.
- Selection of Patients: Ideal candidates are those with comorbid obesity/overweight and a psychiatric disorder linked to reward dysfunction (e.g., BED, SUD, possibly atypical depression with hyperphagia).
- Monitoring: Close monitoring for GI side effects is essential. A collaborative care model with the patient’s primary care physician or endocrinologist is ideal for managing the diabetes or obesity indication and monitoring metabolic parameters.
- The Risk of “Labelling”: It is crucial to frame the treatment around the metabolic comorbidity to avoid stigmatizing the patient’s psychiatric condition or creating unnecessary barriers to insurance coverage.
Conclusion
GLP-1 RAs represent a paradigm-shifting class of drugs whose CNS effects open exciting new avenues for treating reward-based and inflammatory psychiatric disorders. While current use is primarily off-label for comorbid conditions, robust clinical trial data will be necessary to establish their efficacy and safety as primary psychiatric treatments. Psychiatrists must become familiar with their mechanisms, benefits, and risks.
Reference
“GLP-1 Receptor Agonists in Obesity and Psychiatric Disorders” (Frontiers in Neuroscience, 2023).
Give Your feedback: